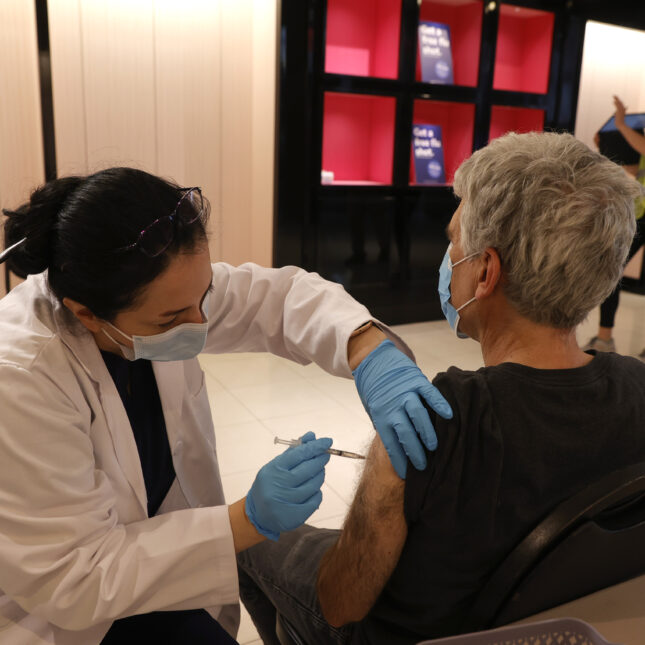
The Food and Drug Administration announced Monday that it had greenlit two updated Covid-19 booster shots in people as young as 6 months old, triggering a process that could see the Moderna and Pfizer-BioNTech vaccines become available in doctors’ offices, clinics, and pharmacies later this week.
The updated approvals relate only to the two messenger RNA vaccines on the U.S. market. An application for an updated vaccine from Novavax is still pending.
“Novavax is currently responding to the FDA’s requests to facilitate final review, and timing is ultimately at the discretion of the FDA,” the company said, noting that doses of its vaccine have arrived in the U.S. for distribution should they receive regulatory clearances.
The updated vaccines must first be recommended by an expert panel that advises the Centers for Disease Control and Prevention on how vaccines should be used. After the Advisory Committee on Immunization Practices votes on whether to recommend the boosters on Tuesday — it is expected to recommend them — CDC Director Mandy Cohen must sign off on the recommendations before the shots can be put into use.
“Vaccination remains critical to public health and continued protection against serious consequences of COVID-19, including hospitalization and death,” said Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, in a statement. “The public can be assured that these updated vaccines have met the agency’s rigorous scientific standards for safety, effectiveness, and manufacturing quality. We very much encourage those who are eligible to consider getting vaccinated.”
These vaccines are also the first Covid shots in this country that were not purchased by the federal government. But most health insurance policies and Medicare will cover the cost of the vaccines. The federal government has put in place a “bridge program,” administered by the CDC, to provide Covid vaccines this fall to roughly 25 million to 30 million adults ages 18 to 64 who either do not have health insurance or whose insurance doesn’t cover Covid shots.
The new boosters from Pfizer and Moderna were approved for people over 12. The FDA also granted an emergency use authorization for anyone over 6 months to receive the boosters.
Pfizer said that it would start distributing the shots now that they are approved, and that the vaccines could be available as soon as the CDC clears their use. That means it is possible shots could be available for some people by the end of the week. Moderna also said the shots would be available in coming days.
“This decision comes at a time when COVID-19 cases are once again climbing. Now, most people 6 months or older in the U.S. are eligible to receive this season’s COVID-19 vaccine, even if they have never been vaccinated against COVID-19 before,” said Albert Bourla, Pfizer’s CEO, in a press release.
“COVID-19 remains a leading cause of death in the U.S. and poses a significant threat to vulnerable populations, particularly as we enter peak respiratory virus season. As the primary circulating strain continues to evolve, updated vaccines will be critical to protecting the population this season,” said Stéphane Bancel, CEO of Moderna, in a statement.
These Covid vaccines are the first that do not include the original strain of the SARS-CoV-2 virus that emerged in Wuhan, China, in late 2019. They are monovalent vaccines — containing a single strain — that target the XBB.1.5 version of Omicron, which was the dominant subvariant when the vaccine strain was selected by the FDA in June.
In recent weeks, it has become apparent a new, highly mutated subvariant, known as BA.2.86, has started to circulate in multiple countries across the globe. The number of mutations this strain includes has raised concern that it could lead to a large spike in cases because those changes may confer on it enhanced capacity to escape the immunity people have developed through vaccination, infection, or a combination of the two.
But it does not appear that the new strain is behind the current increase in cases being seen in the United States. And in a recent risk assessment, the CDC said that while it’s too soon to know what the impact of BA.2.86 will be, it anticipates that the updated Covid vaccines will be effective at preventing severe illness and hospitalizations among people who get vaccinated. Last week Moderna and Pfizer both indicated that testing had shown their updated vaccines do generate strong antibody responses against BA.2.86.
These vaccines can be used as a primary vaccination — a single shot in most cases — or as a booster for people who have been previously vaccinated against Covid.
People older than 5 will be eligible to receive a single dose of the new boosters at least two months after their last dose of Covid vaccine. Children 6 months through 4 years of age who have previously been vaccinated will be eligible to receive one or two doses of the updated vaccine, with timing and number of doses depending on which vaccine they received. (The Moderna shot requires two doses, in total; in younger children, the Pfizer vaccine requires three.)
As things currently stand, the expectation is that for most people — or more precisely, most people who want to get additional doses of Covid vaccine — vaccination will be a once-a-year option, along the model of how flu shots are administered. However, the FDA has indicated that older adults and people who are immunocompromised will be able to get two shots in a year. In April, the FDA followed the lead of health agencies in the United Kingdom and Canada in allowing more vulnerable people to get a second booster to bolster their immunity.
The CDC estimates that roughly 70% of Americans — and nearly 95% of people 65 and older — have had a primary series of Covid vaccinations. But with each successive booster shot campaign, the uptake has shrunk. Only 17% of eligible people got last fall’s bivalent booster. Even among people 65 and older, the figure was only 43%.
Novavax said in a statement that it continues to expect its booster to be “widely available this September for consumers.” The company said that supplies of its vaccine have arrived in the U.S. and are ready to distribute after regulatory approvals are met.
The biggest controversy around the new vaccines is not likely to be the question the FDA had to answer — that is, to whom should these shots be available? It is the one that the CDC and its ACIP committee will answer later in the week: For whom should they be recommended?
Some experts have argued that the boosters are only necessary at this point for those with a high risk of serious disease, because most people have immunity from both previous infection and vaccination. But others argue that a regular booster, given widely, will do the best job of protecting the vulnerable. The debate over which strategy will be most effective is likely to be a major theme of the ACIP meeting.










Exciting news! STAT has moved its comment section to our subscriber-only app, STAT+ Connect. Subscribe to STAT+ today to join the conversation or join us on Twitter, Facebook, LinkedIn, and Threads. Let's stay connected!
To submit a correction request, please visit our Contact Us page.